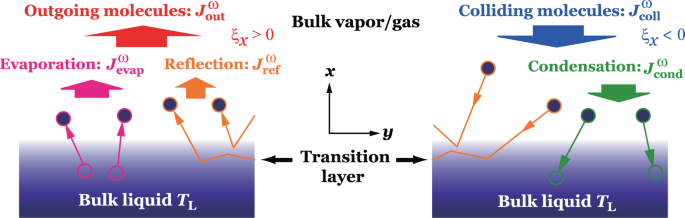
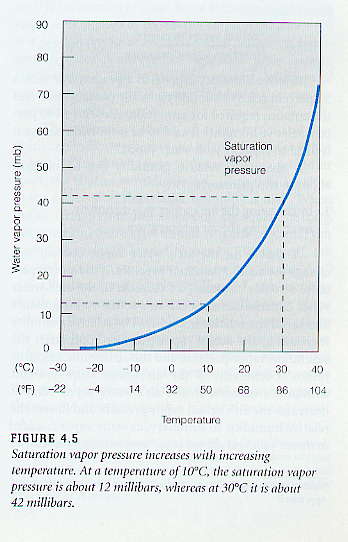
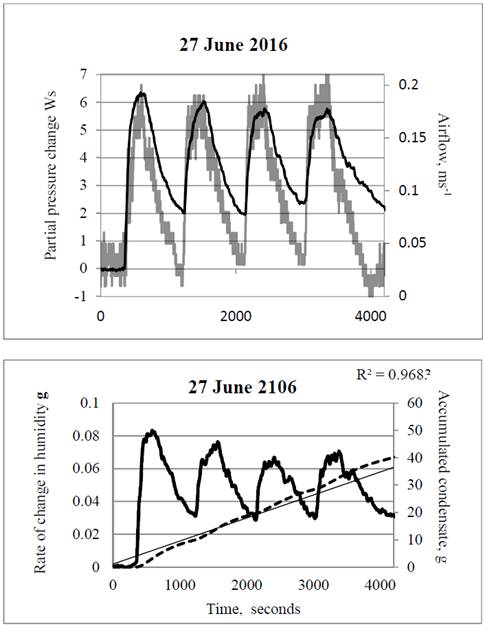
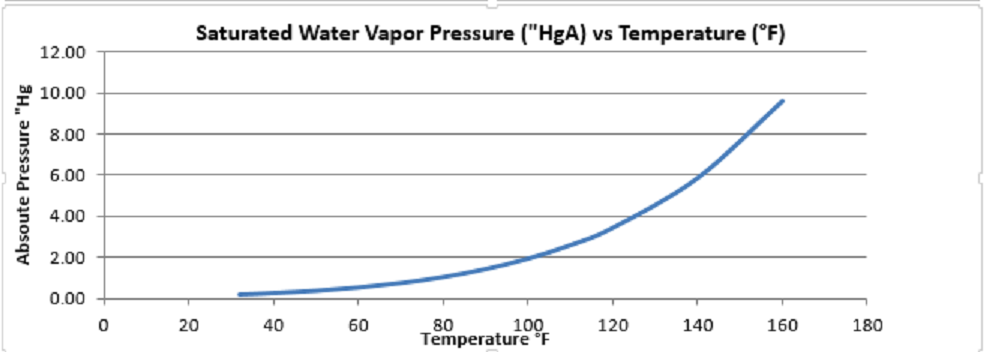
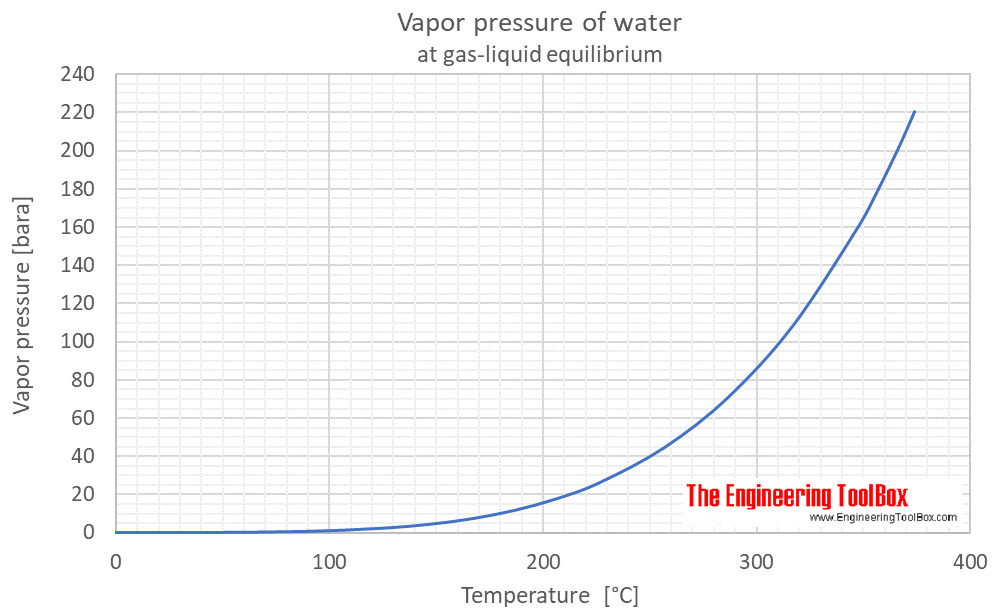
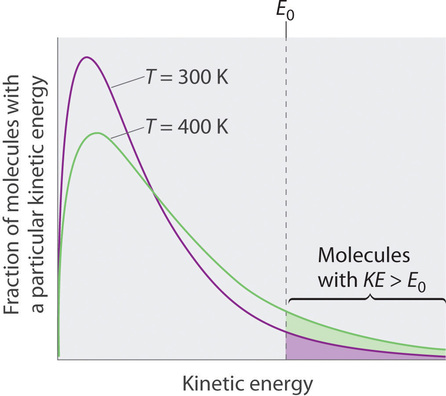
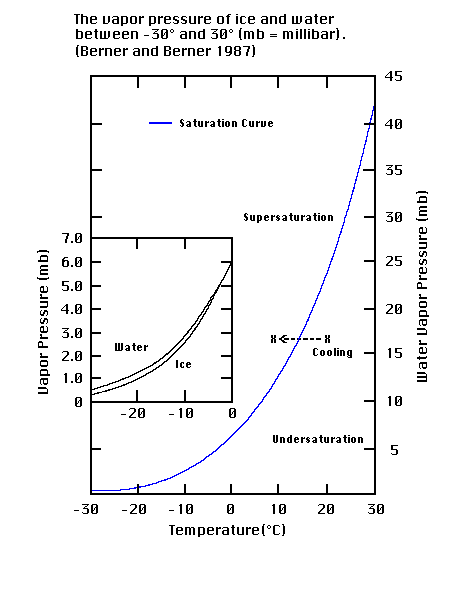
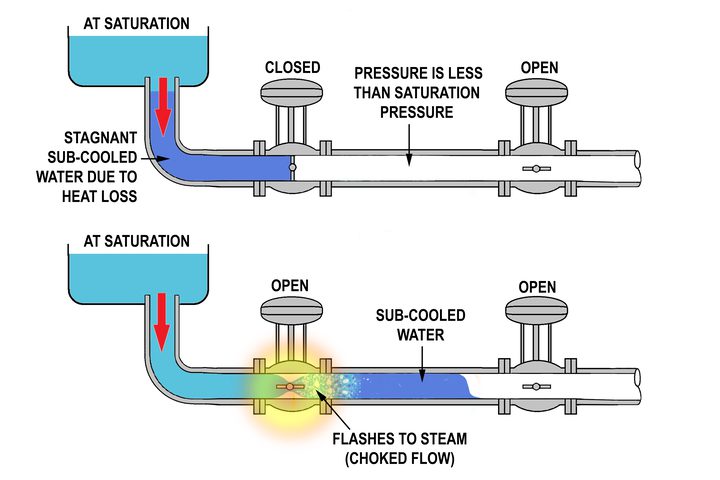
Relative Humidity - Dew Point, Vapor & Partial Pressure, Evaporation, Condensation - Physics - YouTube
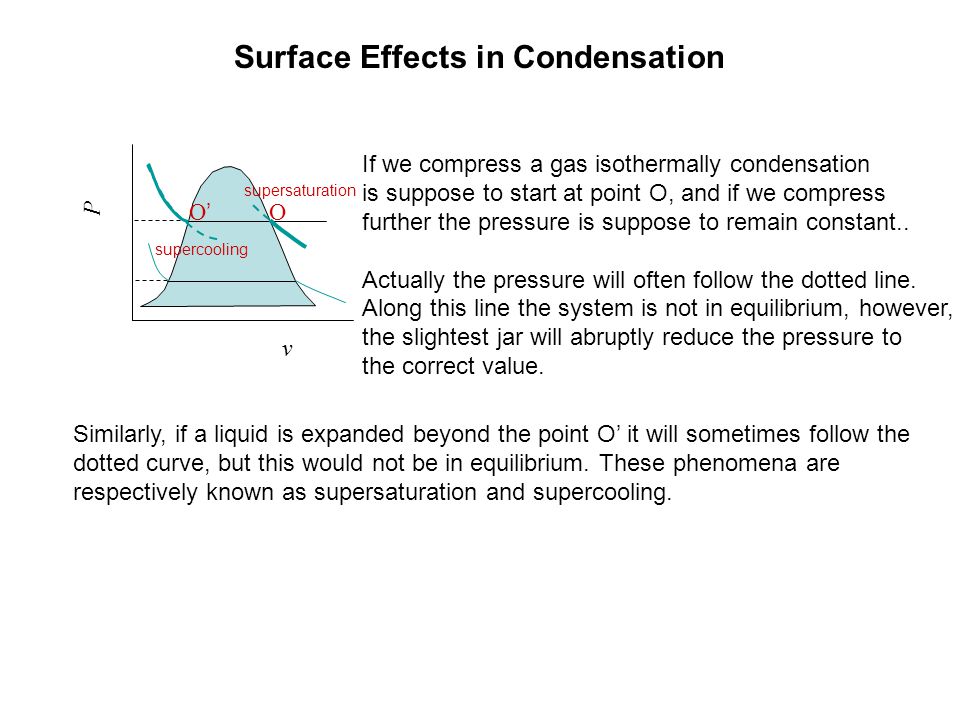
P v Surface Effects in Condensation If we compress a gas isothermally condensation is suppose to start at point O, and if we compress further the pressure. - ppt download

Section 14.2 Vapor Pressure and Boiling Point 1.To understand the relationship among vaporization, condensation and vapor pressure 2.To relate the boiling. - ppt download
Stoichiometry . 10° 20° Temperature C. Fig. 73. 30° In the neighbourhood of the curve of maximum pressure, andalso of the vapour pressure curves for the pure substances, theborder curves are